PERMANENT STAFF
Imma Ratera's lab. Biomaterials and molecular electronics
Imma Ratera's Lab
Biomaterials
a. Molecular bio-interfaces for control environments for cell guidance
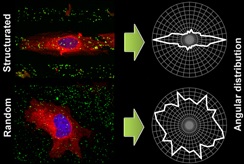
We are interested in the modification of surfaces with molecular and bio-nanomaterials in order to control the cell-material interface for cell guidance studies with many expectations in the field of regenerative medicine and tissue engineering. We have interest in the macroscopic responses of cells when nanoscale profiles made with novel and fully characterized protein nanoparticles (pNPs) are used for the 2D engineering of biological interfaces at the microscale using geometrical patterns by decorating them with SAMs and a modification of the μ-CP technique. We are interested in establishing deep statistical data treatment demonstrating that pNPs-grafted patterned surfaces not only dramatically stimulate cell proliferation but also stimulate cell differentiation aligning and elongating according to the specific patterns (dots, stripes of different sizes). Recently a new technique to deposit nPN in gradients has been used to study and quantify also cell motility.
More recently we are also exploiting the use of electroactive (quinone-hydroquinone) molecular self-assembly monolayers (SAMs) as dynamic, model substrates to trigger the organization of growth factors in a biomimetic way, in order to stimulate not only the spatial but also the temporal cues of the natural microenviroment for cell guidance. Such dynamic molecular bio-interfaces will allow us to organize growth factors in a biomimetic manner being able to understand and control the stimuli that direct cellular behavior towards vascular repair.
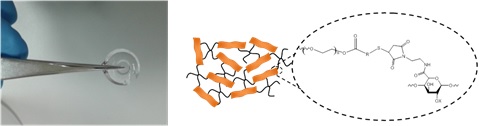
b. Supramolecular hydrogels for cancer immunotherapies (J. Guasch and I. Ratera)
We are also interested on the synthesis, preparation and use of new hydrogels (2D and 3D) for lymphocyte activation, proliferation and differentiation to improve adoptive cell therapy. This therapy is giving impressive results in the treatment of certain blood cancers, achieving remissions of otherwise considered terminal patients. Nevertheless, the ability to expand T cells in high quantities with a determined phenotype and at a reasonable cost remains a limiting factor for the translation of such cancer immunotherapies to public health care systems. In order to improve such limitation, we are developing stimuli-responsive supramolecular hydrogels.